The International System of Units
When you measure something in physics, you need a standard everyone agrees on. Without that, measurements are meaningless. The International System of Units provides exactly this universal framework. Over the centuries, different countries developed their own systems, and three of them became widely used:
- The CGS system (centimeter, gram, and second)
- The MKS system (meter, kilogram, and second)
- The FPS system (foot, pound, and second)
The internationally accepted system today is the Systeme Internationale d’ Unites, abbreviated as SI. You’ll see it called the International System of Units in most textbooks. SI is built on the metric system, which means conversions are based on powers of 10. That makes your calculations far simpler than working with feet and inches. If you’re studying physics at any level, understanding units and measurements starts with knowing the International System of Units inside and out.
History of the International System of Units
The roots of the International System of Units trace back to the French Revolution. In the 1790s, French scientists proposed a decimal-based measurement system that would replace the chaotic patchwork of local units used across Europe. They defined the meter as one ten-millionth of the distance from the North Pole to the equator along a meridian through Paris. The kilogram was defined as the mass of one liter of water at its densest temperature (about 4°C).
France officially adopted the metric system in 1795, making it the first country to do so. Other nations followed slowly. By 1875, the need for international coordination led to the Metre Convention, a treaty signed by 17 nations that established the Bureau International des Poids et Mesures (BIPM) near Paris. The BIPM’s job was to maintain international prototype standards and coordinate measurement science worldwide.
The metric system evolved through several stages. The CGS system (centimeter-gram-second) dominated 19th-century physics, while engineers preferred the MKS system (meter-kilogram-second). In 1960, the 11th General Conference on Weights and Measures (CGPM) formally established the International System of Units as the modern form of the metric system. It included six base units initially. The mole was added as the seventh base unit in 1971.
As of 2026, the BIPM has 64 member states and 36 associate states. The International System of Units is now the official measurement system in every country except the United States, Myanmar, and Liberia, though even these countries use SI in scientific contexts.
Units: Fundamental and Derived
Physical quantities fall into two categories based on how their units are defined. The units for base or fundamental quantities are called fundamental units. You can’t break these down into simpler units. The units of all other physical quantities, which you can express as combinations of fundamental units, are called derived units. This distinction is central to how the International System of Units organizes all of measurement science.
A complete set of fundamental and derived units together is called a system of units.
For any unit to be useful in science, it must satisfy four basic properties:
- It should be readily available and reproducible.
- It must be well-defined, leaving no room for ambiguity.
- It should be invariable, meaning it doesn’t change with time, location, or conditions.
- It should be universally acceptable across all countries and scientific disciplines.
The Seven SI Base Units
The SI system rests on seven fundamental or base units. These are completely independent of each other, and you can express every other physical quantity in terms of these seven. Think of them as the building blocks of all measurement in physics.
| Base Quantity | SI Unit | Symbol | What It Measures |
|---|---|---|---|
| Length | Meter | m | Distance between two points |
| Mass | Kilogram | kg | Amount of matter in an object |
| Time | Second | s | Duration of an event |
| Electric Current | Ampere | A | Flow of electric charge |
| Temperature | Kelvin | K | Thermodynamic temperature |
| Amount of Substance | Mole | mol | Number of elementary entities (atoms, molecules, etc.) |
| Luminous Intensity | Candela | cd | Power of light emitted in a given direction |
Let’s look at each one so you understand exactly what it represents and how it’s defined.
Meter (m)
The meter is the SI unit of length. It’s defined as the distance light travels in a vacuum in exactly 1/299,792,458 of a second. This ties the meter directly to the speed of light, making it one of the most precisely defined units you’ll encounter.
Kilogram (kg)
The kilogram is the SI unit of mass. Until 2019, it was defined by a physical platinum-iridium cylinder kept in a vault in France. Now it’s defined using the Planck constant (h = 6.62607015 x 10-34 J s). This means the kilogram is no longer tied to any physical object, which makes it far more stable and reproducible.
Second (s)
The second is the SI unit of time. It’s defined as the duration of 9,192,631,770 periods of the radiation corresponding to a specific transition of the cesium-133 atom. Atomic clocks based on this definition are so accurate that they would lose less than one second over millions of years.
Ampere (A)
The ampere is the SI unit of electric current. It’s now defined by fixing the elementary charge (e) to exactly 1.602176634 x 10-19 coulombs. In simpler terms, one ampere corresponds to the flow of about 6.24 x 1018 electrons per second past a given point.
Kelvin (K)
The kelvin is the SI unit of thermodynamic temperature. It’s defined by fixing the Boltzmann constant (k) to exactly 1.380649 x 10-23 J/K. Zero kelvin (0 K) represents absolute zero, the lowest temperature theoretically possible, where all molecular motion would cease.
Mole (mol)
The mole is the SI unit for the amount of substance. One mole contains exactly 6.02214076 x 1023 elementary entities. This number is called the Avogadro constant. When you use the mole, you must always specify what entities you’re counting, whether atoms, molecules, ions, or electrons.
Candela (cd)
The candela is the SI unit of luminous intensity. It measures the power of light emitted by a source in a particular direction, weighted by the sensitivity of the human eye. It’s defined by fixing the luminous efficacy of monochromatic radiation of frequency 540 x 1012 Hz to be 683 lumens per watt.
The 2019 SI Redefinition
On 20 May 2019, the SI system underwent its most significant overhaul in decades. Four of the seven base units (kilogram, ampere, kelvin, and mole) were redefined in terms of fixed numerical values of fundamental physical constants. The kilogram was the last unit still defined by a physical artifact, and that changed with this revision.
What this means for you as a student is that every SI base unit is now anchored to an unchanging constant of nature, not to any physical object that could degrade or vary over time. The definitions became more elegant and, more importantly, more precise and universally reproducible.
Here are the four constants that now define these units:
| Constant | Symbol | Exact Value | Defines |
|---|---|---|---|
| Planck constant | h | 6.62607015 x 10-34 J s | Kilogram |
| Elementary charge | e | 1.602176634 x 10-19 C | Ampere |
| Boltzmann constant | k | 1.380649 x 10-23 J/K | Kelvin |
| Avogadro constant | NA | 6.02214076 x 1023 mol-1 | Mole |
Supplementary Units
Apart from the seven base units, the SI system also recognizes two supplementary quantities for measuring angles:
- Plane angle (dθ) is defined as the ratio of the length of arc (ds) to the radius (r). Its unit is the radian (rad).
- Solid angle (dΩ) is defined as the ratio of the intercepted area (dA) of a spherical surface, described about the apex O as the center, to the square of its radius (r2). Its unit is the steradian (sr).
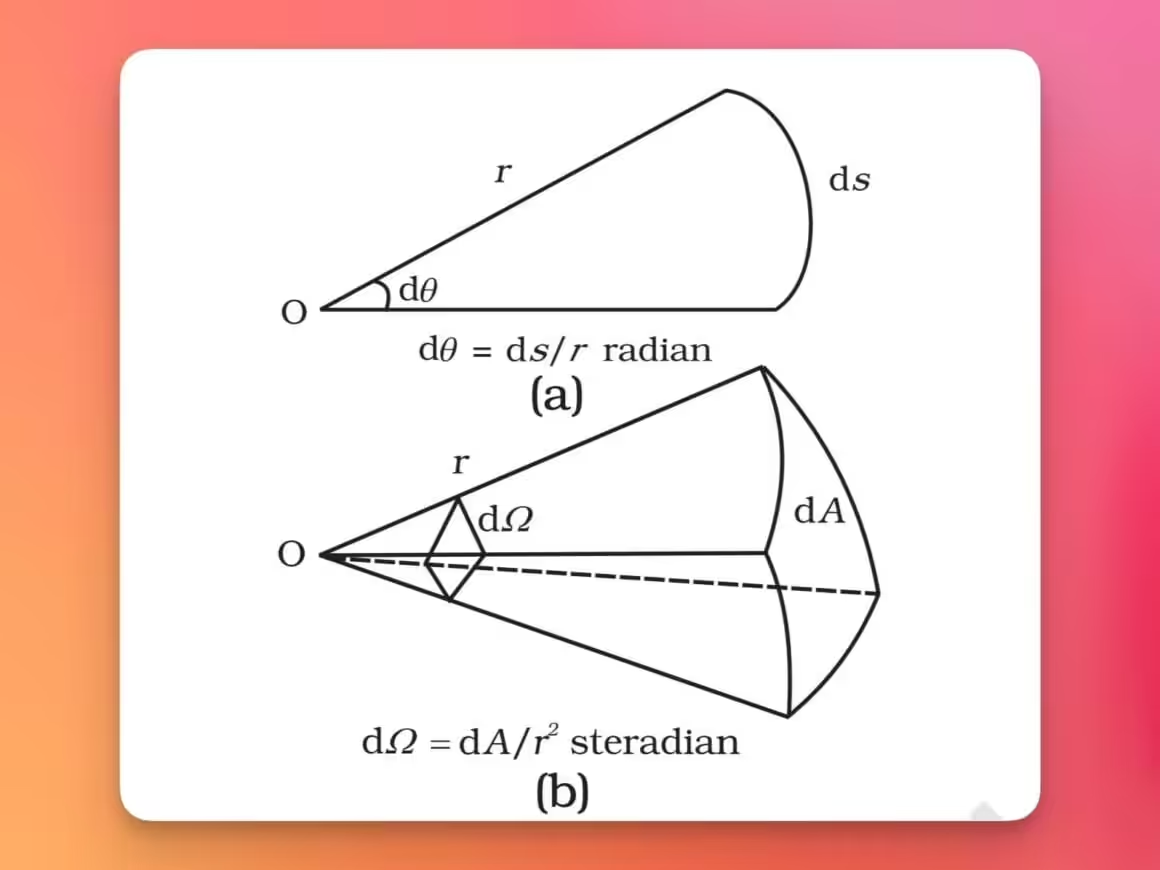
Both the radian and steradian are dimensionless quantities. They were originally classified as “supplementary units” by the SI, but since 1995 they’re treated as derived units with a special status.
Derived Units in the International System of Units
Every physical quantity that isn’t one of the seven base quantities is a derived quantity. Its unit is a derived unit, formed by combining base units through multiplication or division. For example, speed is measured in meters per second (m/s), which combines the base units for length and time.
Some derived units are used so frequently that they get their own names and symbols. The table below lists the most important named derived units you’ll encounter in physics. Each can be traced back to combinations of the seven base units.
| Derived Quantity | SI Unit | Symbol | In Base Units |
|---|---|---|---|
| Force | Newton | N | kg m s-2 |
| Pressure | Pascal | Pa | kg m-1 s-2 |
| Energy / Work | Joule | J | kg m2 s-2 |
| Power | Watt | W | kg m2 s-3 |
| Electric Charge | Coulomb | C | A s |
| Voltage | Volt | V | kg m2 s-3 A-1 |
| Resistance | Ohm | Ω | kg m2 s-3 A-2 |
| Frequency | Hertz | Hz | s-1 |
| Capacitance | Farad | F | kg-1 m-2 s4 A2 |
| Magnetic Flux | Weber | Wb | kg m2 s-2 A-1 |
Whenever you express a derived unit, make sure you can trace it back to the base units. For instance, 1 N = 1 kg m s-2. This skill of dimensional analysis will help you verify equations and catch errors in your calculations. You’ll use this constantly when solving problems involving kinematic equations.
SI Prefixes
The International System of Units uses a set of prefixes to express very large or very small quantities without writing out long strings of zeros. These prefixes represent powers of 10 and attach directly to the unit name. For example, 1 kilometer = 1,000 meters, and 1 milligram = 0.001 grams. Mastering prefixes is essential for working with significant figures and unit conversions.
| Prefix | Symbol | Factor | Example |
|---|---|---|---|
| yotta | Y | 1024 | Mass of Earth (~6 Yg) |
| zetta | Z | 1021 | Global data storage |
| exa | E | 1018 | Age of universe (~0.4 Es) |
| peta | P | 1015 | Distance to nearest stars |
| tera | T | 1012 | 1 THz infrared radiation |
| giga | G | 109 | Computer memory (GB) |
| mega | M | 106 | Radio frequencies (MHz) |
| kilo | k | 103 | 1 km = 1,000 m |
| hecto | h | 102 | Atmospheric pressure (hPa) |
| deca | da | 101 | 1 daL = 10 liters |
| deci | d | 10-1 | 1 dL = 0.1 liter |
| centi | c | 10-2 | 1 cm = 0.01 m |
| milli | m | 10-3 | 1 mg = 0.001 g |
| micro | μ | 10-6 | Bacteria size (~1 μm) |
| nano | n | 10-9 | Semiconductor features (nm) |
| pico | p | 10-12 | Atomic bond lengths |
| femto | f | 10-15 | Nuclear radius (~1 fm) |
| atto | a | 10-18 | Electron orbital period |
| zepto | z | 10-21 | Single molecule mass |
| yocto | y | 10-24 | Proton mass (~1.67 yg) |
In 2022, four new prefixes were added: ronna (R, 1027), quetta (Q, 1030), ronto (r, 10-27), and quecto (q, 10-30). These were the first new prefixes added since 1991 and were driven by the growing need to express extremely large quantities in data science and cosmology.
Real-World Applications of SI Units
SI units aren’t just abstract definitions in a textbook. They’re the backbone of nearly every measurement you’ll encounter in science, engineering, medicine, and daily life. Here’s how different fields depend on the International System of Units.
Science and Research
Every published physics experiment reports results in SI units. CERN measures particle energies in electronvolts (a non-SI unit accepted for use with SI), but all fundamental measurements, from collision energies to detector dimensions, use SI base and derived units. NASA’s Mars Climate Orbiter was famously lost in 1999 because one engineering team used imperial units while another used SI, resulting in a trajectory error that destroyed a $125 million spacecraft.
Engineering and Manufacturing
Modern manufacturing depends on SI units for precision. Semiconductor fabs measure transistor features in nanometers (nm). A single Intel processor has transistors as small as 3 nm. Structural engineers calculate forces in newtons, pressures in pascals, and energy in joules. Without a unified system, global supply chains would be impossible to coordinate.
Medicine and Pharmacology
Drug dosages are prescribed in milligrams (mg) or micrograms (μg). Blood pressure is measured in millimeters of mercury (mmHg, another non-SI unit accepted for medical use), while body temperature uses degrees Celsius (derived from kelvin). Lab results report concentrations in moles per liter (mol/L). A dosage error of even a few milligrams can be the difference between treatment and toxicity, which is why precision in SI units is literally life-or-death in medicine.
Everyday Life
You interact with SI units constantly, even if you don’t think about it. Your phone’s screen brightness is measured in candelas per square meter (cd/m2). The energy content on food labels is in kilojoules (kJ). Your electricity bill is calculated in kilowatt-hours (kWh). Road signs in most countries show distances in kilometers and speed limits in km/h.
When solving physics problems, always convert all quantities to SI base units before plugging them into equations. This prevents unit mismatch errors and ensures your final answer comes out in the correct unit automatically. For example, convert centimeters to meters and grams to kilograms before you start calculating.
Frequently Asked Questions
What does SI stand for and why is it important?
SI stands for Systeme Internationale d’ Unites, or the International System of Units. It’s important because it provides a universal standard for measurement that scientists, engineers, and students across all countries can use consistently. Without it, comparing experimental results or collaborating across borders would be extremely difficult.
How many base units are there in the SI system?
There are seven base units in the SI system: meter (length), kilogram (mass), second (time), ampere (electric current), kelvin (temperature), mole (amount of substance), and candela (luminous intensity). All other units in physics are derived from these seven.
What changed in the 2019 SI redefinition?
In the 2019 redefinition, four base units (kilogram, ampere, kelvin, and mole) were redefined using fixed numerical values of fundamental constants of nature. The kilogram, previously defined by a physical platinum-iridium cylinder, is now defined using the Planck constant. This makes all seven SI base units independent of any physical artifact.
What is the difference between fundamental and derived units?
Fundamental (or base) units are independent units that can’t be expressed in terms of other units. Derived units are combinations of two or more fundamental units. For example, the newton is a derived unit equal to kg m/s², combining the base units for mass, length, and time.
Why is the kilogram no longer defined by a physical object?
The original kilogram prototype, a platinum-iridium cylinder stored in France, was found to have its mass drift very slightly over time due to surface contamination and material changes. By redefining the kilogram using the Planck constant, scientists ensured it would remain perfectly stable and reproducible anywhere in the world without depending on a single physical object.
What are supplementary units in the SI system?
The SI system originally classified two units as supplementary: the radian (for plane angles) and the steradian (for solid angles). Both are dimensionless quantities. Since 1995, they’ve been reclassified as derived units with a special status, but you’ll still see them referred to as supplementary units in many textbooks.
What are SI prefixes and how do they work?
SI prefixes are standardized multipliers based on powers of 10 that attach to unit names. They range from yocto (10⁻²⁴) to yotta (10²⁴), with four newer prefixes (ronto, quecto, ronna, quetta) extending the range further. For example, ‘kilo’ means 10³, so 1 kilometer equals 1,000 meters. Prefixes eliminate the need to write out long strings of zeros.
Which countries don’t use the International System of Units?
The United States, Myanmar, and Liberia are the only countries that haven’t officially adopted the International System of Units as their primary measurement system. However, even in these countries, SI is used in scientific research, military operations, and international trade. The U.S. National Institute of Standards and Technology (NIST) defines all customary units in terms of SI units.