Carbohydrate
Carbohydrates are one of the four major classes of biomolecules, and you’ll encounter them everywhere in biochemistry. From the glucose running through your bloodstream right now to the cellulose holding up every tree you’ve ever seen, these molecules are fundamental to life. Primarily produced by plants, carbohydrates form the most extensive group of naturally occurring organic compounds. Common examples include cane sugar, glucose, and starch. They are compounds of carbon, hydrogen, and oxygen, and are also known as saccharides because their basic components are sugars.
In this study note, you’ll learn about the general formula of carbohydrates, the three major classes (monosaccharides, oligosaccharides, and polysaccharides), and the structural and biological significance of key sugars like glucose, fructose, sucrose, starch, and cellulose.
General Formula
Most carbohydrates have a general formula, Cx(H2O)y, and were considered to be hydrates of carbon (that’s where the term “carbohydrate” comes from). For example, the molecular formula of glucose (C6H12O6) readily fits into this general formula: C6(H2O)6.
However, you shouldn’t rely on this formula alone to classify a compound. Acetic acid (CH3COOH) fits into the general formula C2(H2O)2 but is not a carbohydrate. Similarly, rhamnose (C6H12O5) is a carbohydrate but does not meet this criterion.
After observing a large number of their reactions, scientists deduced that carbohydrates contain specific functional groups. Chemically, they may be defined as optically active polyhydroxy aldehydes or ketones or the compounds that produce such units on hydrolysis.
Certain carbohydrates, which are sweet in taste, are also known as sugars. The common table sugar you use at home is called sucrose, whereas the sugar present in milk is lactose. On the basis of their behavior on hydrolysis, carbohydrates are broadly divided into three different groups, which we’ll discuss in detail below.
Remember the three classes of carbohydrates by their size: monosaccharides are single sugar units (glucose, fructose), oligosaccharides are short chains of 2-9 units (sucrose, lactose), and polysaccharides are long chains of hundreds or thousands of units (starch, cellulose). The prefix tells you the size: mono = one, oligo = few, poly = many.
Monosaccharides
A carbohydrate that cannot be hydrolyzed further to yield simpler units of polyhydroxy aldehydes or ketones is known as a monosaccharide. Think of monosaccharides as the building blocks of all carbohydrates, one of the four major classes of biomolecules. Around twenty monosaccharides are known to exist in nature. They are composed of 3-7 carbon atoms and are known as biomicromolecules. Examples include glucose, fructose, and ribose.
Monosaccharides are further classified based on the number of carbon atoms and the functional group present in them. Monosaccharides containing an aldehyde group are known as aldoses and those containing a keto group are known as ketoses. The number of carbon atoms constituting the monosaccharide is subsequently introduced in its name too, as shown below:
| Carbon atoms | General term | Aldehyde | Ketone |
| 3 | Triose | Aldotriose | Ketotriose |
| 4 | Tetrose | Aldotetrose | Ketotetrose |
| 5 | Pentose | Aldopentose | Ketopentose |
| 6 | Hexose | Aldohexose | Ketohexose |
| 7 | Heptose | Aldoheptose | Ketoheptose |
Monosaccharides may be modified chemically to form several types of derived monosaccharides. Deoxy sugars are a good example: deoxygenation (removal of oxygen from the second carbon) of ribose produces deoxyribose, which is an important constituent of DNA.
Other derived monosaccharides include amino sugars (glucosamine, N-acetyl glucosamine), sugar acids (ascorbic acid, glucuronic acid), and sugar alcohols (mannitol).
Let’s now discuss certain monosaccharides in more detail.
Glucose
Biologically and medically, glucose is arguably the most important monosaccharide in existence. You’ll find it freely in nature as well as in combined form. It is abundant in honey and sweet fruits such as ripe grapes. It is also found in your blood and is thus known as “blood sugar”.
Structure of glucose
Also known as dextrose, glucose is chemically an aldohexose. It serves as the monomeric unit of many larger carbohydrates such as starch and cellulose. Its structure was determined by chemists based on the following observations:
- The molecular formula of glucose was deduced to be C6H12O6.
- On prolonged heating with hydrogen iodide, it forms n-hexane. This tells you that all the six carbon atoms in glucose are linked in a straight chain.
- Glucose reacts with hydroxylamine to form an oxime. On the addition of a molecule of hydrogen cyanide, it produces cyanohydrin. These reactions confirm the presence of a carbonyl group (>C = O) in glucose.
- Glucose gets oxidized to a carboxylic acid-containing six carbon atoms (gluconic acid) in reaction with mild oxidizing agents such as bromine water. This indicates that the carbonyl group present in glucose is an aldehydic group.
- On acetylation with acetic anhydride, glucose produces glucose pentaacetate. This confirms the presence of five -OH groups in the structure of glucose. Since it exists as a stable compound, you can infer that these -OH groups are all attached to different carbon atoms.
- On oxidation with nitric acid, both glucose and gluconic acid yield a dicarboxylic acid known as saccharic acid. This indicates the presence of a primary alcoholic group (-OH) in glucose.
The exact spatial arrangement of the different -OH groups in glucose was given by Emil Fischer after a careful study of many other properties of the sugar. Glucose is correctly named as D(+)-glucose. The ‘D’ here represents the configuration in three-dimensional space whereas the ‘(+)’ represents the dextrorotatory nature of the molecule.
Cyclic structure of glucose
Certain facts could not be explained by the linear structure of glucose, as follows:
- Although it has an aldehyde group, glucose does not give Schiff’s test and also does not form the hydrogensulphite addition product with NaHSO3.
- The pentaacetate of glucose does not react with hydroxylamine. This indicates the absence of a free aldehyde group.
- Glucose is found to exist in two different crystalline forms in solution, known as the α-form and the β-form.
The α-form of glucose (melting point: 419 K) is obtained by crystallization from a concentrated solution of glucose at 303 K. Similarly, the β-form (melting point: 423 K) is obtained by crystallization from a hot and saturated aqueous solution of glucose at 371 K.
Scientists were unable to explain this behavior by the open chain structure of glucose. As a result, they proposed that one of the -OH groups may possibly add to the -CHO group and form a cyclic hemiacetal structure.
Later, it was discovered that glucose forms a six-membered ring in which the -OH at C-5 is involved in the ring formation process. This explains the absence of a free -CHO group and also the existence of glucose in two different crystalline forms. Both of these cyclic hemiacetal forms exist in equilibrium with the open-chain structure of glucose.
The two cyclic forms of glucose differ only in the configuration of the hydroxyl group at C-1, known as the anomeric carbon (the aldehyde carbon just before cyclization). Such isomers are known as anomers.
The six-membered cyclic structure of glucose is known as the pyranose structure, in analogy with a cyclic organic compound called pyran. Pyran is a cyclic organic compound with one oxygen atom. Diagrammatically, the cyclic form of glucose is accurately represented by the Haworth structure.
Biological significance
Glucose is the main source of energy for your body and is carried by the bloodstream to all your body parts. The normal concentration of glucose in blood is 80-100 mg per 100 mL of blood. It is oxidized by living cells to produce energy in the form of ATP.
Since glucose requires no digestion, it may be given intravenously to patients who cannot take food orally. Glucose is found in the urine of patients who have diabetes mellitus. The condition in which glucose is excreted in urine is known as glycosuria.
Fructose
Fructose is one of the most important ketohexoses in nature. It is obtained along with glucose during the hydrolysis of the disaccharide, sucrose. You’ll find it abundantly in fruits, honey, and various vegetables. In its pure form, it is used as a sweetener in the food industry.
Like glucose, fructose has the molecular formula C6H12O6. Scientists observed its reactions and found out that it contains a ketone group at C-2 and six carbon atoms in a straight chain, as in the case of glucose. It belongs to D-series as well, but unlike glucose, it is laevorotatory in nature. Thus, it is appropriately written as D-(-)-fructose.
Fructose also exists in two different cyclic forms that are obtained by the addition of -OH at C-5 to the carbonyl group. The ring thus formed is a five membered ring and is known as the furanose structure, with analogy to a five-membered cyclic compound called furan.
Fructose is the major constituent of the polysaccharide inulin, a polysaccharide present in many plants such as dahlia tubers, chicory roots, and Jerusalem artichokes. It is the sweetest of all natural sugars, being about twice as sweet as glucose.
Galactose
Galactose is an aldohexose and occurs, along with glucose, in lactose and in many other oligosaccharides and polysaccharides such as pectin, gums, and mucilage. Galactose is an epimer of glucose, differing only in the spatial arrangement of the -H and -OH groups around C-4.
Galactose is also synthesized in the mammary glands to produce the lactose in milk. It is a constituent of glycolipids (carbohydrate-lipid complexes) and glycoproteins in many cell membranes, such as those found in nervous tissues. It is less than half as sweet as glucose.
The inability of infants to metabolize galactose is an inherited condition known as galactosemia. In this disorder, the galactose concentration increases noticeably in the blood and also appears in the urine. Galactosemia results in vomiting, diarrhea, hepatomegaly, and often mental retardation.
If not recognized within a few days after birth, it can even lead to death. However, if the diagnosis is made early and lactose is removed from the diet, the symptoms disappear and the normal growth of the child may be resumed.
Oligosaccharides
Oligosaccharides are small carbohydrates formed by the condensation of 2-9 monosaccharide units. Condensation is the chemical combination of two molecules to form one molecule with the loss of a small molecule, usually water. Oligosaccharides are considered biomacromolecules.
The monosaccharide units are joined together by a glycosidic bond between the aldehyde or ketone group of one monosaccharide and the alcohol group of another. Depending on the number of monosaccharide molecules condensed to form an oligosaccharide, they can be:
- Disaccharides (sucrose, lactose, maltose, trehalose)
- Trisaccharides (raffinose – composed of glucose, fructose, and galactose)
- Tetrasaccharides (stachyose)
Let’s study some important oligosaccharides in detail.
Sucrose
Sucrose is the common table sugar used in sweets, drinks, ice creams, cakes, and many more food items. It is commercially obtained from sugarcane stems and the roots of the sugar beet plant. On hydrolysis, it produces an equimolar mixture of D-(+)-glucose and D-(-) fructose.
These two monosaccharide units are held together by a glycosidic linkage between C-1 of α-D-glucose and C-2 of β-D-fructose. Sucrose is dextrorotatory in nature, but after hydrolysis it yields dextrorotatory glucose and laevorotatory fructose.
Because the laevorotation of fructose (-92.4 degrees) is much more than the dextrorotation of glucose (+52.5 degrees), the resulting mixture is laevorotatory in nature. Thus, the hydrolysis of sucrose brings about a change in the sign of rotation from dextrorotatory (+) to laevorotatory (-). As a result, the solution is known as invert sugar.
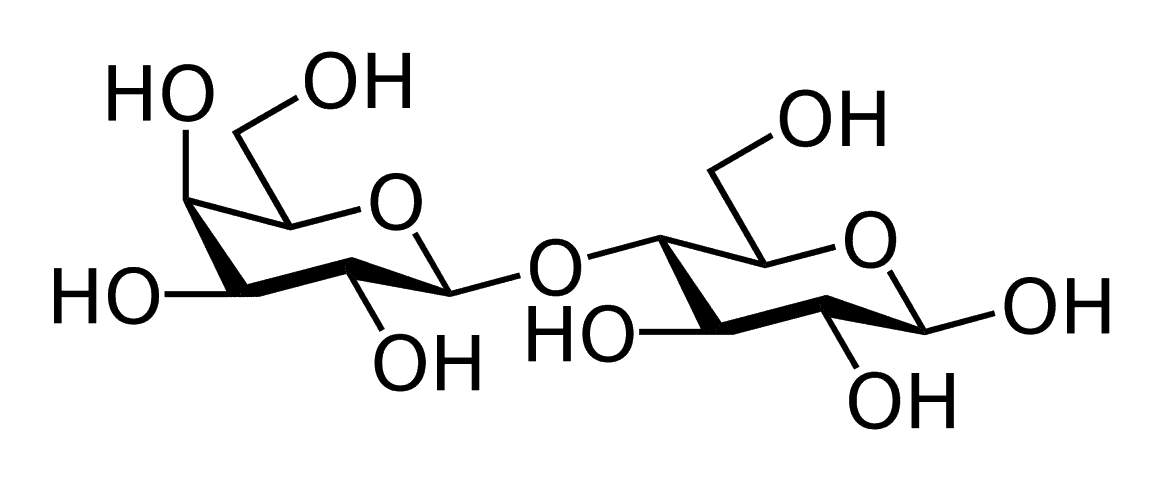
Maltose
Maltose or malt sugar is another disaccharide that is composed of two α-D-glucose units, in which C-1 of one glucose unit is linked to C-4 of another glucose unit.
Lactose
Lactose is also known as milk sugar because of its prominent presence in milk. It is composed of β-D-galactose and β-D-glucose. The glycosidic linkage is between C-1 of galactose and C-4 of glucose.
Reducing Sugars
Reducing sugars are sugars that can reduce Cu2+ ions to Cu+ ions. This property is found in all saccharides that possess free aldehyde or ketone groups, including all monosaccharides. On the basis of this property, Fehling’s and Benedict’s tests are used to detect the presence of glucose in urine.
When it comes to disaccharides, sucrose (which contains glucose and fructose) is a non-reducing sugar because both the aldehyde group of glucose and the ketone group of fructose participate in the formation of the glycosidic bond between the two of them. However, some other disaccharides such as maltose and lactose have reducing groups.
Polysaccharides
Polysaccharides are complex carbohydrates that contain a large number of monosaccharide units joined together by glycosidic linkages. These are by far the most commonly encountered carbohydrates in nature. They are mainly used by organisms for two purposes: food storage and structural support.
Let’s now look at the structure of some important polysaccharides found in nature.
The type of glycosidic linkage determines whether a polysaccharide stores energy or provides structural support. Alpha (α) linkages produce coiled, easily digestible chains (starch, glycogen). Beta (β) linkages produce straight, rigid chains that resist digestion (cellulose, chitin). This single structural difference explains why you can digest a potato but not a piece of wood.
Starch
Starch [(C6H10O5)n] is the most important storage polysaccharide found in the plant kingdom. It is mainly concentrated in seeds, roots, and tubers. Corn, wheat, potatoes, rice, and cassava are the main dietary sources of starch. It is a polymer of α-glucose and consists of two components: amylose (13-20%) and amylopectin (80-87%).
Amylose is the water-soluble component of starch that constitutes about 15-20% of its bulk. It is a long, unbranched chain with 200-1000 α-D-(+)-glucose units held together by α-1,4 glycosidic linkage.
Amylopectin is a water-insoluble and branched polymer of α-D-glucose units, in which the chain is formed by α-1,4 glycosidic linkage whereas branching takes place by α-1,6 glycosidic linkage. There are about 24-30 glucose residues between two branch points.
Despite the presence of numerous polar -OH groups, starch molecules are insoluble in water because of their very large size. However, it does form colloidal dispersions in hot water that give an intense blue-black color with free iodine because starch can hold I2 molecules in its secondary helices. You can use a starch solution to detect free iodine or a dilute iodine solution to detect starch.
You can easily convert starch into glucose by heating it with water and a little acid (such as hydrochloric or sulphuric acid). It is also readily hydrolyzed at room temperature by certain digestive enzymes such as amylase and maltase.
Glycogen
Glycogen is the main energy-storage carbohydrate of the animal kingdom. It is formed by the polymerization of glucose (a process known as glycogenesis) and is stored abundantly in the liver and in muscle tissues.
Structurally, it is quite similar to the amylopectin fraction of starch, except that it is more highly branched. There are 12-18 glucose residues between two branch points (α-1,6 glycosidic linkages) in a glycogen molecule.
Muscle glycogen granules (β-particles) are spherical in shape and contain up to 60,000 glucose residues. In the liver, there are similar granules and also rosettes of glycogen granules that seem to be aggregated β-particles.
Cellulose
Cellulose is the most abundant organic substance found in nature. It is the main structural component of plants and wood. Cotton fibers are composed of more or less pure cellulose, whereas dry wood consists of around 50% cellulose. Cellulose plays a vital role in the textile and paper industries.
Like starch and glycogen, cellulose is a polymer of glucose. However, cellulose differs from the other two in that the glucose units in cellulose are joined by β-1,4 glycosidic linkages instead of α-1,4 glycosidic linkages. The stereochemistry of the β-anomer allows cellulose to form an extended chain that can form hydrogen bonds with adjacent cellulose molecules. The large number of hydrogen bonds so formed contributes to the strength of the resulting plant cell walls.
Cellulose has a much greater resistance to hydrolysis compared to starch or glycogen. It is not appreciably hydrolyzed when boiled in a 1% sulphuric acid solution. It does not show any color reaction with iodine.
You cannot digest cellulose because your body has no enzymes capable of catalyzing its hydrolysis. There is only a little bacterial metabolism of cellulose in the human colon. However, some microorganisms found in soil and in the digestive tracts of certain animals produce enzymes that do catalyze the breakdown of cellulose. The presence of these microorganisms explains why cows and other herbivorous animals can live on grass and why termites feed on wood.
Other Polysaccharides
Inulin is a polysaccharide of fructose found in tubers and roots of dahlias, artichokes, and dandelions. It is highly soluble in water and is used to determine the glomerular filtration rate. However, since it is not hydrolyzed by intestinal enzymes, it has no nutritional value.
Dextrins are intermediates formed during the hydrolysis of starch.
Chitin is a structural polysaccharide found in the exoskeleton of crustaceans, insects, and fungi. It consists of N-acetyl-D-glucosamine units joined by β-1,4 glycosidic bonds. Pectin is a partially methylated polysaccharide found in fruits. It is a polymer of galacturonic acid residues linked with α-1,4 glycosidic bonds with some galactose and/or arabinose branches.
Glycosaminoglycans or mucopolysaccharides form a part of the connective tissue. These polymers act as shock absorbers between bones and prevent them from rubbing against each other when you move. Osteoarthritis, the most common form of arthritis, occurs due to the loss of glycosaminoglycans at joints.
Complex polysaccharides are found on the surfaces of almost all cells, often linked to proteins to form glycoproteins. They serve as “labels” or antigens that allow organisms to distinguish their own cells from foreign substances, materials, or pathogens. Antigen recognition forms the basis of a very important biochemical principle: molecular shape carries information that guides the reactions of life.
Polysaccharides found on the surface of your red blood cells give rise to different blood types, which are commonly classified by the ABO system.
Frequently Asked Questions
What are carbohydrates and what are they made of?
Carbohydrates are optically active polyhydroxy aldehydes or ketones, or compounds that produce such units on hydrolysis. They are composed of carbon, hydrogen, and oxygen. Most carbohydrates follow the general formula Cx(H2O)y, though this formula alone is not sufficient to classify a compound as a carbohydrate. They are also known as saccharides and are primarily produced by plants through photosynthesis.
What is the difference between monosaccharides, oligosaccharides, and polysaccharides?
Monosaccharides are the simplest carbohydrates that cannot be hydrolyzed further into simpler sugars. Examples include glucose and fructose. Oligosaccharides are formed by the condensation of 2-9 monosaccharide units joined by glycosidic bonds (e.g., sucrose, lactose). Polysaccharides contain a large number of monosaccharide units and serve as energy storage (starch, glycogen) or structural components (cellulose, chitin).
Why does glucose exist in a cyclic form?
Glucose exists in a cyclic form because the -OH group at C-5 reacts with the aldehyde group (-CHO) at C-1 to form a six-membered cyclic hemiacetal structure. This cyclic form explains why glucose does not give Schiff’s test (no free aldehyde group) and why it exists in two crystalline forms known as the α-anomer and the β-anomer, which differ in the configuration of the hydroxyl group at the anomeric carbon (C-1).
What is the difference between starch and cellulose?
Both starch and cellulose are polymers of glucose, but they differ in the type of glycosidic linkage. Starch uses α-1,4 glycosidic linkages (and α-1,6 for branching in amylopectin), while cellulose uses β-1,4 glycosidic linkages. This structural difference means humans can digest starch using enzymes like amylase, but cannot digest cellulose because we lack the necessary enzymes. Starch functions as an energy storage molecule in plants, whereas cellulose serves as a structural component in plant cell walls.
What are reducing and non-reducing sugars?
Reducing sugars are sugars that can reduce Cu2+ ions to Cu+ ions because they have a free aldehyde or ketone group. All monosaccharides are reducing sugars. Among disaccharides, maltose and lactose are reducing sugars. Sucrose is a non-reducing sugar because both the aldehyde group of glucose and the ketone group of fructose are involved in the glycosidic bond, leaving no free reducing group. Fehling’s and Benedict’s tests are used to detect reducing sugars.
What is invert sugar and how is it formed?
Invert sugar is the equimolar mixture of glucose and fructose obtained by the hydrolysis of sucrose. Sucrose itself is dextrorotatory (rotates plane-polarized light to the right). However, when hydrolyzed, it produces dextrorotatory glucose (+52.5 degrees) and laevorotatory fructose (-92.4 degrees). Since the laevorotation of fructose is greater than the dextrorotation of glucose, the resulting mixture is laevorotatory. This change in the sign of optical rotation from positive to negative is called inversion, hence the name invert sugar.