Raman Effect – Biography of CV Raman, Theory and Examples
This is a complete guide to the Raman Effect, covering CV Raman’s life, the theory behind Raman Scattering, and practical applications of Raman Spectroscopy.
If you’re a physics student (graduate or undergraduate), this article will give you everything you need to understand Raman’s groundbreaking work. We’ll cover the history, the classical theory with full derivations, and why this discovery still matters today. For a deeper dive with downloadable resources, check out our complete The Raman Effect: Theory, Discovery, and Applications product.

About CV Raman
Sir Chandrasekhara Venkata Raman (known as CV Raman) was an Indian physicist, mathematician, and Nobel laureate. His first name was Venkata. He was a Tamil Brahmin and the second of eight children.
Raman was born on 7th November 1888 at Thiruvanaikaval, near Tiruchirappalli in what was then Madras Presidency (now Tamil Nadu). His father, Chandrasekhar Iyer, lectured in mathematics and physics at a local college, which sparked Raman’s early interest in science. His mother, Parvathi Ammal, came from a family of Sanskrit scholars.
Here’s how his remarkable career unfolded:
- Raman moved to Visakhapatnam at a young age, where he studied at St. Aloysius Anglo-Indian High School. He passed his matriculation exam at just 11 years old and earned his B.A. degree from Presidency College, Madras, at 16.
- After graduating with an M.A. in physics (finishing first in his class), he joined the Indian government’s finance department in 1907 as an Assistant Accountant General. But his passion for physics never faded — he spent evenings and weekends conducting experiments at the Indian Association for the Cultivation of Science (IACS) in Kolkata.
- In 1917, at just 28 years old, Raman resigned from government service to become the Palit Professor of Physics at the University of Calcutta. He took a significant pay cut to pursue science full-time.
- On 28th February 1928, Raman announced his discovery of the Raman Effect through his experiments on light scattering. This date is now celebrated as National Science Day in India.
- In 1930, Raman received the Nobel Prize in Physics for this discovery, becoming the first Asian to win a Nobel Prize in science. He was also the first person to receive the Nobel Prize while working entirely outside Europe and North America.
- In 1932, Raman and S. Bhagavantam discovered the quantum photon spin, further confirming the quantum nature of light.
- In 1934, Raman founded the Indian Academy of Sciences and later established the Raman Research Institute in Bangalore in 1948, where he continued research until his death on 21st November 1970.
History of the Raman Effect
Before 1928, scientists had to use infrared spectroscopy to study the vibrational and rotational properties of molecules. This was inconvenient and limited in scope. Infrared detectors were primitive, sample preparation was tedious, and many important molecules (like O2 and N2) simply didn’t show up in infrared spectra at all. Raman’s discovery changed everything. Suddenly, researchers could excite molecules using visible light and study their properties with far greater ease.
The theoretical groundwork for the Raman Effect was laid before Raman’s experiments. Adolf Smekal predicted inelastic light scattering in 1923 based on quantum mechanical arguments. Kramers and Heisenberg expanded on this work in 1925 with their dispersion theory, followed by Dirac in 1927 with his quantum radiation theory. But it was Raman and his student K.S. Krishnan who first observed experimental evidence of inelastic scattering in liquids in 1928.
The story of the discovery itself is worth telling. During a voyage to Europe in 1921, Raman was struck by the deep blue color of the Mediterranean Sea. Lord Rayleigh had attributed the sea’s color to reflection of the sky, but Raman wasn’t convinced. He carried a simple prism and a miniature spectroscope on the ship and demonstrated that the sea’s color was due to molecular scattering of light by water molecules. This observation set him on the path that would lead to his greatest discovery seven years later.
Raman immediately recognized he was dealing with something fundamental. The phenomenon was analogous to the Compton Effect (where X-rays scatter off electrons with a change in wavelength), but this was happening with visible light and molecules. While the Compton Effect involved free or loosely bound electrons, Raman scattering involved the internal vibrations and rotations of entire molecules.
To confirm his discovery, Raman used a mercury arc lamp and a spectrograph to record the spectrum of scattered light. The equipment was remarkably simple by today’s standards — sunlight focused through a telescope served as his first light source, and he used complementary light filters to isolate the scattered wavelengths.
What Raman observed was striking. When any transparent substance (solid, liquid, or gas) was illuminated by the mercury arc, the scattered light contained more than just the original frequencies. The spectrum showed new lines, bands, and continuous radiation shifted from the incident frequencies. The unmodified radiation was Rayleigh scattering. The modified radiation was something new entirely — a direct window into the vibrational and rotational energy states of molecules.
What is the Raman Effect?
The Raman Effect is the change in wavelength of light when a light beam is deflected by molecules.
When a beam of light passes through a transparent, dust-free sample, most of the light continues in its original direction. But a small portion scatters in other directions. Most of this scattered light has the same wavelength as the incident beam (this is Rayleigh scattering). However, a tiny fraction has a different wavelength. This wavelength shift is the Raman Effect.
Think of it this way. When photons from the light source collide with molecules in the sample, most collisions are elastic — the photons bounce off with the same energy and frequency they had before. But occasionally, a collision is inelastic. The molecule either absorbs energy from the photon (leaving the photon with less energy and a lower frequency) or gives energy to the photon (leaving the photon with more energy and a higher frequency). The scattered photon leaves with a different frequency than it arrived with.
These frequency changes tell us about the energy transitions happening inside the molecule. They reveal information about vibrational and rotational states that would otherwise be difficult to measure. Every molecule has a unique set of vibrational modes — think of them as the molecule’s natural frequencies of oscillation — and the Raman spectrum captures these like a molecular fingerprint.
The Raman Effect is weak. For a typical liquid, the intensity of the modified light might be only 1/100,000th of the incident beam. But every molecule produces a characteristic pattern of Raman lines, with intensity proportional to the number of scattering molecules. This makes Raman spectroscopy useful for both qualitative and quantitative analysis.
The energies corresponding to Raman frequency shifts match the energies of vibrational and rotational transitions within molecules. For simple gaseous molecules, you can observe pure rotational shifts (with smaller energy changes). In liquids, molecular rotation is hindered by intermolecular interactions, so you won’t see discrete rotational Raman lines. The Raman Effect primarily reveals vibrational transitions, which produce observable shifts in solids, liquids, and gases alike.
Gases at ordinary temperatures have low molecular concentration and produce weak Raman effects. That’s why most Raman spectroscopy work focuses on solids and liquids. Modern laser sources have made gas-phase Raman measurements more practical, but the signal remains inherently weaker.
Raman Scattering Explained
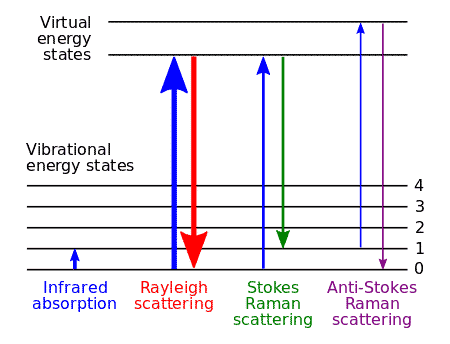
When light of a specific frequency passes through any substance (gas, liquid, or solid), the scattered light contains more than just the original frequency. Raman discovered that light scattered at right angles includes frequencies that are generally lower (sometimes higher) than the incident light.
This phenomenon is Raman Scattering. The scattered lines with lower frequencies are called Stokes lines — named after George Stokes, who first described fluorescence. Those with higher frequencies are anti-Stokes lines.
If \( f \) is the frequency of incident light and \( f’ \) is the frequency of a scattered line, then \( f – f’ \) is the Raman Frequency. This frequency difference doesn’t depend on the incident light frequency. It’s a constant that characterizes the substance itself. You could use green, blue, or red laser light and the Raman shift would remain the same — only the absolute frequencies of the scattered lines would change.
A remarkable feature of Raman Scattering is that Raman Frequencies match (within experimental error) the frequencies obtained from infrared rotation-vibration spectra of the same substance. This confirmed that both techniques probe the same molecular energy levels, just through different physical mechanisms.
Physically, Raman scattering occurs because of inelastic collisions between photons and the molecular electron cloud. The energy difference between incident and scattered photons corresponds to a quantum of vibrational or rotational energy that the molecule either absorbs or releases.
At thermal equilibrium, most molecules occupy the ground vibrational state (following the Boltzmann distribution). That’s why Stokes lines are always more intense than anti-Stokes lines at room temperature — more molecules are available to absorb energy than to give it up. The ratio of anti-Stokes to Stokes intensity can actually be used as a molecular thermometer to measure sample temperature.
Advantages of Raman Spectroscopy
Raman spectroscopy offers several advantages over infrared spectroscopy:
- Works across all phases. It works for gases, liquids, and solids. Infrared spectra of liquids and solids are often too diffuse to be quantitatively useful because of strong absorption and broad bands.
- Detects non-polar molecules. Non-polar molecules like \( \text{O}_2 \), \( \text{N}_2 \), and \( \text{Cl}_2 \) show Raman scattering even though they don’t appear in infrared spectra. This is because Raman activity requires a change in polarizability, not a permanent dipole moment.
- Unique access to non-polar vibrations. Rotation-vibration changes in non-polar molecules can only be observed through Raman spectroscopy. This makes it indispensable for studying symmetric molecules.
- Measurement convenience. Raman frequencies are only slightly different from incident frequencies. By choosing appropriate incident radiation, you can bring the scattered lines into the visible region where measurement is easier. Infrared measurements require specialized detectors and optics that are transparent to IR radiation.
- Simpler experimental setup. It uses visible or ultraviolet radiation instead of infrared, which means you can use glass optics, fiber optic probes, and standard CCD detectors. Infrared spectroscopy requires specialized materials like KBr, ZnSe, or diamond for windows and lenses.
- Water-compatible. Water is a weak Raman scatterer but a strong infrared absorber. This makes Raman spectroscopy ideal for studying aqueous solutions, biological samples, and in vivo measurements — situations where infrared spectroscopy struggles.
- Non-destructive and requires minimal sample preparation. Samples can be measured through glass containers, plastic packaging, or even at a distance using focused laser beams. No grinding, pressing into pellets, or dissolving is needed.
Applications of Raman Spectroscopy
Raman spectroscopy has evolved from a laboratory curiosity into one of the most versatile analytical techniques in modern science. Its non-destructive nature, minimal sample preparation, and ability to work through containers make it invaluable across multiple fields:
- Biology and medicine. Investigating biological systems, including polypeptides, proteins, and nucleic acids in aqueous solutions. Water doesn’t interfere with Raman measurements the way it does with infrared. Surface-Enhanced Raman Spectroscopy (SERS) has pushed detection limits to the single-molecule level, opening doors to cancer diagnostics and glucose monitoring.
- Molecular structure determination. The Raman spectrum provides a “fingerprint” of the molecule. Symmetric vibrations that are weak or absent in IR spectra often produce strong Raman signals, making the two techniques complementary for complete structural analysis.
- Pharmaceutical analysis. Used to identify compounds, verify drug purity, detect counterfeit medications, and monitor crystallization processes in real-time. The FDA has approved Raman-based methods for process analytical technology (PAT) in pharmaceutical manufacturing.
- Materials science. Characterization of carbon nanotubes, graphene, and other nanomaterials. The G-band and D-band in carbon materials’ Raman spectra reveal information about structural quality, number of layers, and defect density that no other technique can provide as easily.
- Art conservation and archaeology. Non-destructive analysis of pigments, binders, and degradation products in paintings, manuscripts, and artifacts. Portable Raman spectrometers can identify materials on-site without removing samples from priceless artworks.
- Forensics and security. Identifying unknown substances at crime scenes, detecting explosives and narcotics through sealed containers, and analyzing fibers, paints, and inks. Spatially Offset Raman Spectroscopy (SORS) can probe through opaque layers like packaging or skin.
- Environmental monitoring. Real-time detection of pollutants in water, identification of microplastics, and monitoring of atmospheric gases. Raman lidar systems can profile atmospheric composition at distances of several kilometers.
- Semiconductor industry. Measuring stress, strain, composition, and crystal quality in semiconductor wafers. Micro-Raman mapping can reveal stress distributions around device structures with sub-micron spatial resolution.
Classical Theory of Raman Effect
The classical theory of the Raman Effect (also called the polarizability theory) was developed by George Placzek in 1934. It provides an elegant explanation for why Raman scattering occurs.
The basic idea is this: when a photon interacts with a molecule, it causes the electrons and nuclei to move, inducing an oscillating electric dipole. This dipole then radiates photons, some of which have different frequencies from the incident light. The key insight is that this frequency shift happens only when the molecular vibration changes the molecule’s polarizability.
The electric field \( E \) of electromagnetic radiation induces a dipole moment \( \mu \) in a molecule:
$$ \mu = \alpha E \quad \text{…(1)} $$
Here, \( \alpha \) is the polarizability of the molecule — a measure of how easily the electron cloud can be distorted by an external electric field. Larger, more loosely held electron clouds have higher polarizability. The electric field oscillates as:
$$ E = E_0 \sin \omega t = E_0 \sin 2\pi \nu t \quad \text{…(2)} $$
where \( E_0 \) is the amplitude and \( \nu \) is the frequency of the incident light.
Substituting equation (2) into equation (1):
$$ \mu = \alpha E_0 \sin 2\pi \nu t \quad \text{…(3)} $$
An oscillating dipole at frequency \( \nu \) radiates light at that same frequency. This is Rayleigh scattering — elastic scattering with no energy transfer. But here’s where it gets interesting.
If the polarizability changes during molecular vibration, we can write:
$$ \alpha = \alpha_0 + \frac{d\alpha}{dq} q \quad \text{…(4)} $$
where \( \alpha_0 \) is the equilibrium polarizability and \( q \) describes the molecular vibration coordinate (the displacement from equilibrium). The vibration itself oscillates as:
$$ q = q_0 \sin 2\pi \nu_m t \quad \text{…(5)} $$
Here, \( q_0 \) is the amplitude and \( \nu_m \) is the molecular vibration frequency.
Combining equations (4) and (5):
$$ \alpha = \alpha_0 + \frac{d\alpha}{dq} q_0 \sin 2\pi \nu_m t \quad \text{…(6)} $$
Substituting this into equation (3):
$$ \mu = \alpha_0 E_0 \sin 2\pi \nu t + \frac{d\alpha}{dq} q_0 E_0 \sin 2\pi \nu t \sin 2\pi \nu_m t \quad \text{…(7)} $$
Using the trigonometric identity \( \sin x \sin y = \frac{1}{2}[\cos(x-y) – \cos(x+y)] \), this becomes:
$$ \mu = \alpha_0 E_0 \sin 2\pi \nu t + \frac{1}{2} \frac{d\alpha}{dq} q_0 E_0 \left[ \cos 2\pi(\nu – \nu_m)t – \cos 2\pi(\nu + \nu_m)t \right] \quad \text{…(8)} $$
This result is the heart of the classical Raman theory. It shows that the oscillating dipole has three frequency components:
- The incident frequency \( \nu \) with amplitude \( \alpha_0 E_0 \) — this is Rayleigh scattering. It’s the strongest component because it depends on the full equilibrium polarizability.
- The frequency \( \nu – \nu_m \) — this is the Stokes line. The scattered photon has lost energy equal to one quantum of molecular vibration. The molecule moves from the ground vibrational state to an excited state.
- The frequency \( \nu + \nu_m \) — this is the Anti-Stokes line. The scattered photon has gained energy from the molecule. The molecule was already in an excited vibrational state and drops to the ground state.
The Stokes and anti-Stokes lines have much smaller amplitudes of \( \frac{1}{2} \frac{d\alpha}{dq} q_0 E_0 \), which explains why Raman scattering is so much weaker than Rayleigh scattering. The ratio depends on how strongly the polarizability changes with vibration — a quantity determined by the molecule’s electronic structure.
If the molecular vibration doesn’t change the polarizability (meaning \( \frac{d\alpha}{dq} = 0 \)), then the dipole oscillates only at the incident frequency. No Raman scattering occurs.
This gives us the selection rule for Raman activity: for a vibration or rotation to be Raman-active, it must cause a change in molecular polarizability:
$$ \frac{d\alpha}{dq} \neq 0 \quad \text{…(9)} $$
This is why homonuclear diatomic molecules like \( \text{H}_2 \), \( \text{N}_2 \), and \( \text{O}_2 \) show Raman spectra but no infrared spectra. They lack permanent dipole moments (so no IR absorption), but their vibrations do change polarizability (so Raman scattering occurs). The changing polarizability induces a changing dipole moment at the vibrational frequency, which radiates the Raman-shifted light.
This complementarity between IR and Raman activity is formalized in the mutual exclusion rule: for molecules with a center of symmetry, vibrations that are Raman-active are IR-inactive, and vice versa. This rule is particularly useful for determining molecular symmetry — if the same vibration appears in both the IR and Raman spectra, the molecule cannot have a center of inversion.
Further Reading
Want a comprehensive, downloadable resource? Our The Raman Effect: Theory, Discovery, and Applications covers everything in this article and more, with additional worked examples and practice problems.
Frequently Asked Questions
Who was CV Raman?
Sir Chandrasekhara Venkata Raman (1888–1970) was an Indian physicist and mathematician who won the 1930 Nobel Prize in Physics. He discovered the Raman Effect, which describes how light changes wavelength when scattered by molecules. He was the first Asian and first non-European/non-American to win a Nobel Prize in science while working entirely in his home country.
What was CV Raman’s first name?
Venkata was his first name. “CV Raman” stands for Chandrasekhara Venkata Raman. Chandrasekhara was his family name (from his father Chandrasekhar Iyer).
When was CV Raman born?
He was born on 7th November 1888 at Thiruvanaikaval, near Tiruchirappalli in the Madras Presidency (now Tamil Nadu, India). He passed away on 21st November 1970 in Bangalore at the age of 82.
When did CV Raman receive the Nobel Prize?
Raman received the Nobel Prize in Physics in 1930 for his work on light scattering and the discovery of the Raman Effect. He was the first Asian scientist to win a Nobel Prize in any science field. The Nobel committee specifically cited “his work on the scattering of light and for the discovery of the effect named after him.”
Why is National Science Day celebrated on 28th February?
National Science Day commemorates the announcement of the Raman Effect on 28th February 1928. It marks the day Raman publicly revealed his discovery to the scientific community, not his birthday (which was 7th November). The Indian government designated this day as National Science Day in 1986 to encourage scientific awareness.
What is the Raman Effect?
The Raman Effect is the change in wavelength of light when it’s scattered by molecules. Most scattered light keeps the same wavelength (Rayleigh scattering), but a small fraction — roughly 1 in 10 million photons — shifts to different wavelengths. This shift reveals information about molecular vibrations and rotations, acting as a molecular fingerprint unique to each substance.
What is spectroscopy?
Spectroscopy is the study of how electromagnetic radiation interacts with matter. Scientists use it to identify substances and understand their properties by analyzing the spectrum of absorbed, emitted, or scattered radiation. Different types include absorption spectroscopy (IR, UV-Vis), emission spectroscopy (fluorescence), and scattering spectroscopy (Raman).
What is the polarizability theory?
The polarizability theory is the classical explanation for the Raman Effect, developed by George Placzek in 1934. It explains that Raman scattering occurs when molecular vibrations cause changes in the molecule’s polarizability (the ease with which the electron cloud can be distorted). When polarizability changes during vibration, the induced dipole radiates at shifted frequencies — producing Stokes and Anti-Stokes lines.
What are Stokes and anti-Stokes lines?
Stokes lines are scattered light with lower frequency (longer wavelength) than the incident light — the molecule absorbs energy from the photon. Anti-Stokes lines have higher frequency (shorter wavelength) because the molecule gives energy to the photon. At room temperature, Stokes lines are more intense because more molecules start in the ground vibrational state (following the Boltzmann distribution). The intensity ratio of anti-Stokes to Stokes lines can be used to measure temperature.
What is the mutual exclusion rule?
The mutual exclusion rule states that for molecules with a center of symmetry (like CO2 or benzene), vibrations that are Raman-active are infrared-inactive, and vice versa. If a vibration appears in both IR and Raman spectra, the molecule lacks a center of inversion. This rule is a powerful tool for determining molecular symmetry.