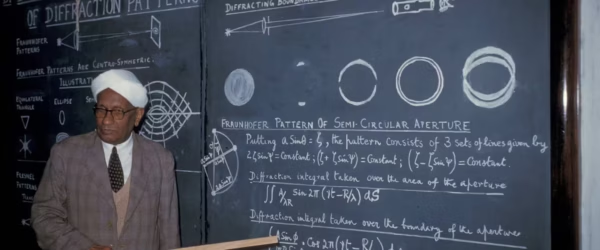
Raman Effect – Biography of CV Raman, Theory and Examples
This complete guide covers everything about the Raman Effect. You'll learn about CV Raman's life, the theory behind Raman Scattering,…
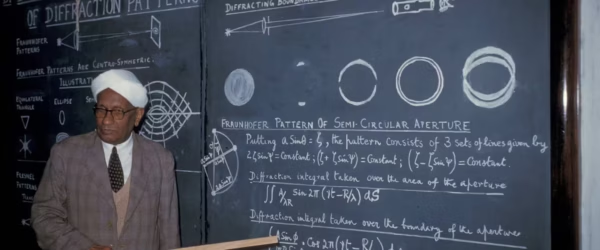
This complete guide covers everything about the Raman Effect. You'll learn about CV Raman's life, the theory behind Raman Scattering,…

Unimolecular reactions seem impossible. A single molecule just reacts on its own? Where does the energy come from? Lindemann solved…