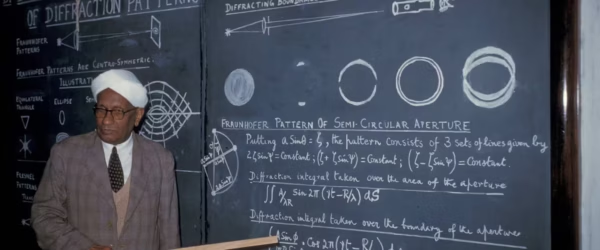
Raman Effect – Biography of CV Raman, Theory and Examples
This complete guide covers everything about the Raman Effect. You'll learn about CV Raman's life, the theory behind Raman Scattering,…
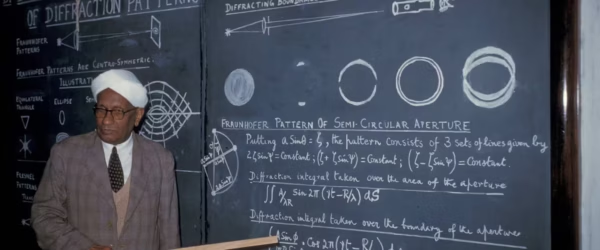
This complete guide covers everything about the Raman Effect. You'll learn about CV Raman's life, the theory behind Raman Scattering,…
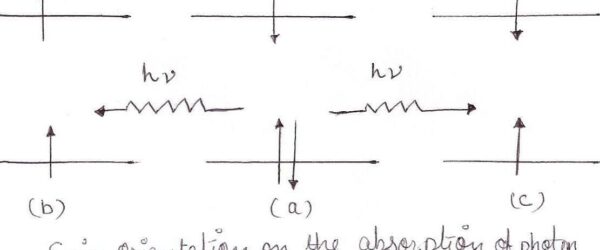
The Jablonski diagram maps what happens to a molecule after it absorbs light. Fluorescence, phosphorescence, internal conversion, intersystem crossing —…