
15+ Best Physical Chemistry Books for Undergraduates
Physical chemistry sits at the intersection of math and chemistry, making it notoriously difficult to study from a bad textbook.…
Chemistry related articles by Gaurav Tiwari to help you excel in NEET, JEE, XI, XII, B. Sc., M. Sc., MS and PhD. These articles are precise and are explained with graphs, pictures and videos to help you understand these better.

Physical chemistry sits at the intersection of math and chemistry, making it notoriously difficult to study from a bad textbook.…

Most inorganic chemistry textbooks feel like they were written to punish you. Dense notation, zero context, and explanations that assume…
Organic chemistry nearly ended my academic confidence. I remember staring at reaction mechanisms wondering if I'd ever understand them. The…

Biomolecules are the chemical compounds that make life possible. Proteins, carbohydrates, lipids, nucleic acids, and enzymes each play critical roles…

A chemistry degree from a top university opens doors to research, pharmaceuticals, environmental science, and academia. I've researched the best…

Chemistry YouTube channels have transformed how students learn the subject. From organic chemistry reaction mechanisms to physical chemistry derivations, the…

Chemical reactions are everywhere. From the rust on your car to the food you digest, every physical process involves a…
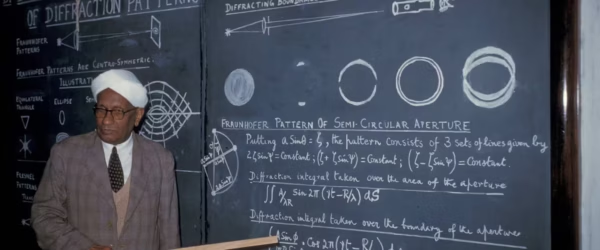
This complete guide covers everything about the Raman Effect. You'll learn about CV Raman's life, the theory behind Raman Scattering,…
Class 12 chemistry is weird. One chapter you're memorizing transition metal colors. The next you're balancing organic reaction mechanisms that…

Chemistry notes can make or break your understanding of the subject. Good notes distill complex reactions and concepts into reviewable…
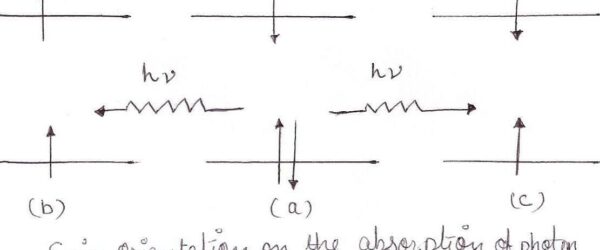
The Jablonski diagram maps what happens to a molecule after it absorbs light. Fluorescence, phosphorescence, internal conversion, intersystem crossing —…

Unimolecular reactions seem impossible. A single molecule just reacts on its own? Where does the energy come from? Lindemann solved…